Ocean Acidification (Week 12) - Post 1
Ocean Acidification - Notes
 Coral-
Coral-
-Ocean Acidification Demonstration-
-Ocean Acidification-
"PMEL CO2 - Carbon Dioxide Program." Ocean Carbon Storage. N.p., n.d. Web. 06 May 2018.
-Ocean Acidification, or 'OA' for short, is the term given to the chemical changes in the ocean as a result of carbon emissions.
-What is Ocean Acidification-
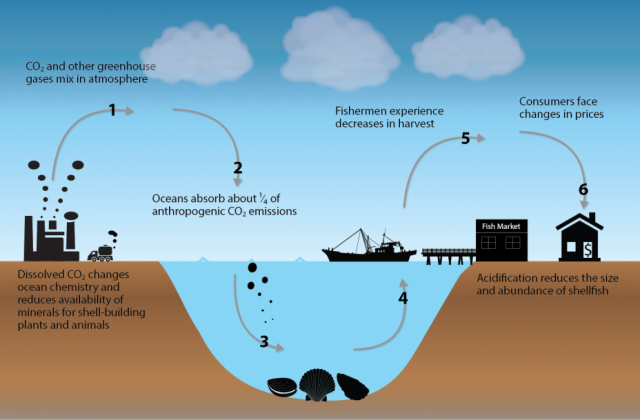
-Fundamental changes in seawater chemistry are occurring throughout the
world's oceans. Since the beginning of the industrial revolution, the
release of carbon dioxide (CO2) from humankind's industrial and agricultural activities has increased the amount of CO2 in the atmosphere. The ocean absorbs about a quarter of the CO2 we release into the atmosphere every year, so as atmospheric CO2
levels increase, so do the levels in the ocean. Initially, many
scientists focused on the benefits of the ocean removing this greenhouse
gas from the atmosphere. However, decades of ocean observations now
show that there is also a downside — the CO2 absorbed by the ocean is changing the chemistry of the seawater, a process called ocean acidification.
Chemistry-
When carbon dioxide (CO2) is absorbed by seawater, chemical
reactions occur that reduce seawater pH, carbonate ion concentration,
and saturation states of biologically important calcium carbonate
minerals. These chemical reactions are termed "ocean acidification" or
"OA" for short. Calcium carbonate minerals are the building blocks for
the skeletons and shells of many marine organisms. In areas where most
life now congregates in the ocean, the seawater is supersaturated with
respect to calcium carbonate minerals. This means there are abundant
building blocks for calcifying organisms to build their skeletons and
shells. However, continued ocean acidification is causing many parts of
the ocean to become undersaturated with these minerals, which is likely
to affect the ability of some organisms to produce and maintain their
shells.
Since the beginning of the Industrial Revolution, the pH of surface
ocean waters has fallen by 0.1 pH units. Since the pH scale, like the
Richter scale, is logarithmic, this change represents approximately a 30 percent increase in acidity.
Future predictions indicate that the oceans will continue to absorb
carbon dioxide, further increasing ocean acidity. Estimates of future
carbon dioxide levels, based on business as usual emission scenarios,
indicate that by the end of this century the surface waters of the ocean
could have acidity levels nearly 150 percent higher, resulting in a pH
that the oceans haven’t experienced for more than 20 million years.
-Biological Impacts-
Ocean acidification is expected to impact ocean species to varying
degrees. Photosynthetic algae and seagrasses may benefit from higher CO2 conditions in the ocean, as they require CO2
to live just like plants on land. On the other hand, studies have shown
that lower environmental calcium carbonate saturation states can have a
dramatic effect on some calcifying species, including oysters, clams,
sea urchins, shallow water corals, deep sea corals, and calcareous
plankton. Today, more than a billion people worldwide rely on food from
the ocean as their primary source of protein. Thus, both jobs and food
security in the U.S. and around the world depend on the fish and
shellfish in our oceans.
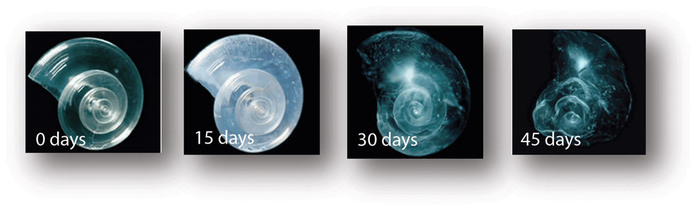
Pterpods-
The pteropod, or “sea butterfly”, is a tiny sea creature about the size
of a small pea. Pteropods are eaten by organisms ranging in size from
tiny krill to whales and are a food source for North Pacific juvenile
salmon. The photos below show that a pteropod’s shell dissolves over 45
day when placed in sea water with pH and carbonate levels projected for
the year 2100
 Coral-
Coral-
Many marine organisms that produce calcium carbonate shells or skeletons are negatively impacted by increasing CO2
levels and decreasing pH in seawater. For example, increasing ocean
acidification has been shown to significantly reduce the ability of
reef-building corals to produce their skeletons. In a recent paper,
coral biologists reported that ocean acidification could compromise the
successful fertilization, larval settlement and survivorship of Elkhorn
coral, an endangered species. These research results suggest that ocean
acidification could severely impact the ability of coral reefs to
recover from disturbance. Other research indicates that, by the end of
this century, coral reefs may erode faster than they can be rebuilt.
This could compromise the long-term viability of these ecosystems and
perhaps impact the estimated one million species that depend on coral
reef habitat.
“NOAA Ocean Acidification Demonstration.” YouTube, 26 Feb. 2010, youtu.be/xuttOKcTPQs.
Key Concepts:
+The chemistry of the ocean is dependent on the chemistry of the atmosphere (More CO2 in the atmosphere leads to more CO2 dissolved in the ocean)
+More CO2 dissolved in the ocean makes the ocean more acidic.
+Changes in ocean acidity impact marine organisms that have calcium carbonate shells
+More CO2 dissolved in the ocean makes the ocean more acidic.
+Changes in ocean acidity impact marine organisms that have calcium carbonate shells
-Acidic ocean water is corrosive to all calcium carbonate shells and skeletons
-Effects of Ocean and Coastal Acidification on Ecosystem-
https://www.epa.gov/ocean-acidification/effects-ocean-and-coastal-acidification-ecosystems
Ocean life is very diverse, and increased acidity can harm or help
individual plant and animal species in different ways. It may not be
obvious to us at first, but some organisms are likely to become more
abundant, and others less so. For example, seagrasses may grow faster if
more dissolved carbon dioxide is available, while the number of oysters
may decrease as fewer larvae complete their life cycle due to increased
acidity. In this way, scientists expect ocean and coastal acidification
to affect entire ecosystems
including one animal at the top of the food chain— humans. Humans rely
on the ocean for food and other economic resources. Ocean and coastal
acidification may not just affect life underwater, but ultimately all of
us.
Human economies rely on ecosystem services provided by healthy oceans and coasts, but ocean and coastal acidification threaten these services. For example, each year in the United States, oysters, clams and scallops supply seafood valued at 400$ million. Unless serious steps are taken to halt ocean and coastal acidification, a falling supply of shellfish is estimated to lead to consumer losses of roughly 480$ million per year by the end of the century. The risk is even greater for societies that have traditionally relied on the bounty of the sea, as exemplified by the traditional fisheries of U.S. Native American tribes.
https://climate.nasa.gov/nasa_science/science/
http://nasawavelength.org/resource-search?facetSort=1&educationalLevel=High+school
https://www.esrl.noaa.gov/gmd/ccgg/trends/monthly.html
https://www.pmel.noaa.gov/co2/
Comments
Post a Comment